Where Vietnamese Ambition Meets Japanese Precision
26 January 2026
As part of Vietnam’s comprehensive healthcare digital transformation under Resolution No. 20/NQ-TW and Decision No. 749/QD-TTg of the Government, electronic prescriptions and national pharmaceutical data interconnection have become mandatory requirements across the healthcare system.
In reality, antimicrobial resistance in Vietnam has reached an alarming level, prompting the Ministry of Health to strengthen regulatory control through centralized data systems. Electronic prescribing is therefore no longer optional—it is now a legal prerequisite for healthcare facilities to operate sustainably from 2025 onward.
This article provides a complete overview of the implementation roadmap, penalties, registration procedures, and supporting digital solutions, helping clinic owners, pharmacies, doctors, and pharmacists comply proactively and avoid unexpected inspections, insurance claim rejections, or administrative penalties.
Deadline: before October 1, 2025
Applicable entities:
Hospitals with 300 beds or more
Must implement electronic prescribing and interconnect prescription data with the National Prescription System.
Retail drug outlets (pharmacies and drug counters)
Must dispense prescription medicines using electronic prescription codes via their management software and transmit sales data to the national system.
Important note:
Merely installing software is not sufficient. Facilities must actively transmit real transaction data. Any non-interconnected transactions will be deemed non-compliant.
Deadline: before January 1, 2026
Applies to all healthcare facilities other than Tier 1, 2, and 3 hospitals, including:
General and specialist clinics
Dental clinics and aesthetic clinics
Traditional medicine facilities
Mandatory requirements:
100% of prescriptions must be issued electronically
Applies to prescription drugs, OTC medicines, traditional medicine, and biological products
Regardless of patient volume or prescription frequency

Under Decree No. 117/2020/ND-CP, multiple violations may be penalized simultaneously during a single inspection.
Real case in Phu Tho Province:
A private clinic was fined for three common violations:
Failure to submit mandatory healthcare activity reports
Improper prescription practices (handwritten prescriptions, missing information, incorrect formats)
Failure to issue electronic prescriptions and interconnect data
👉 Penalty: VND 3,000,000 per violation
👉 Total fine: VND 9,000,000 in one inspection
According to Point d, Clause 4, Article 59, the act of:
“Dispensing prescription drugs without a valid prescription”
👉 Penalty: VND 10,000,000 for the first violation
Case study in Lam Dong Province:
A pharmacy chain was inspected across 10 outlets in a single day, and all 10 outlets were fined, resulting in a substantial total penalty.
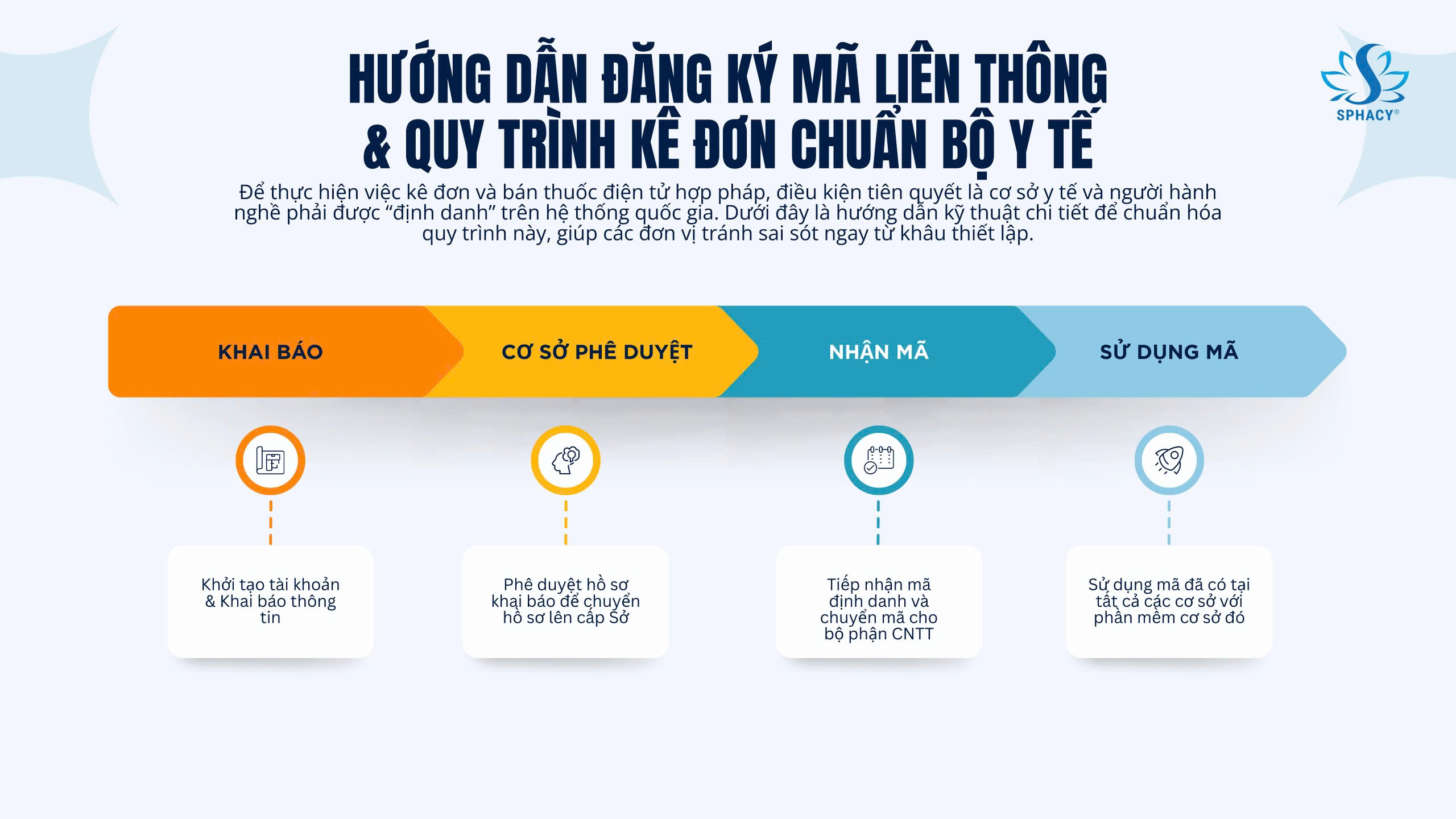
Step 1:
Access donthuocquocgia.vn → Register healthcare facility → Complete information form
Step 2:
Upload legal documents (PDF format):
List of licensed healthcare professionals
Operating license of the healthcare facility
Step 3:
Approval by the Department of Health → Issuance of interconnection code → Configuration within the facility’s HIS software
One lifelong identification code per practitioner
Valid across all workplaces (public hospitals, private clinics, after-hours practices)
When changing workplaces, only an information update is required—no new code issuance
Prescription code: 14 characters (in accordance with Circulars No. 04/2022 and 27/2021)
Prescription validity:
Standard prescriptions: 5 days
Chronic disease prescriptions: 60–90 days (mandatory notation required)
Patient identification:
A phone number is mandatory to enable electronic delivery of the prescription to the patient
Currently approved by the Ministry of Health:
54 healthcare facility management software systems
66 pharmacy management software systems
Facilities should verify that their current software is included in the approved list.
Through a cooperation program with SPHACY Joint Stock Company, the following support is provided:
Free trial of pharmaceutical company and pharmacy management software
Full integration with the National Pharmaceutical Database, including built-in e-invoicing and digital signatures, with continuous updates in compliance with the latest tax regulations, circulars, and official guidance
Facilities are advised to use a dedicated business bank account
Prevents tax risks arising from mixed personal and business cash flows
Strictly prohibited:
Selling medicines on general platforms such as Shopee, Lazada, Facebook, or Zalo
Livestream sales of medicines
Misleading or exaggerated advertising claims
Permitted:
Sales on Ministry of Health–licensed pharmaceutical e-commerce platforms
Sales via the pharmacy’s official website or proprietary mobile app
Mandatory storage of consultation records for at least 24 months

Electronic prescribing and national pharmaceutical data interconnection are not merely administrative requirements—they represent a fundamental transformation toward transparency and sustainability in Vietnam’s healthcare system.
Early compliance before 2026 enables healthcare facilities to:
Avoid administrative fines of up to VND 10,000,000 per violation
Prevent health insurance claim rejections
Enhance credibility and patient volume